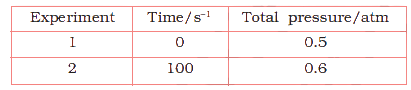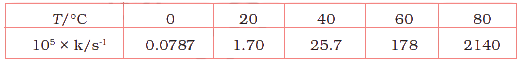Bhattacharjee Sir is truly an exceptional chemistry teacher whose passion for the subject and dedication to his students make learning an inspiring experience. His deep knowledge of both theoretical and practical aspects of chemistry transforms every lesson into an engaging journey of discovery. He has a remarkable ability to simplify complex chemical equations and concepts, making them understandable and enjoyable for students of all levels. His teaching style combines clarity, patience, and real-life examples that spark curiosity and encourage critical thinking. He goes beyond textbooks, motivating students to explore experiments and develop a genuine love for the subject. Bhattacharjee Sir’s approachable nature and willingness to answer every question create a positive, supportive classroom environment. His guidance has helped countless students achieve academic success and develop confidence in chemistry. With his passion and commitment, Bhattacharjee Sir stands out as an inspiring mentor who leaves a lasting impact on his students’ educational journeys.