Hybridisation is a hypothetical concept which explains about the formation and structure of molecules.
Defination - the intermixing of atomic orbitals to produce symmetrical low energy containg hybrid orbitals is know as hybridisation.
There are following types of hybdridisations are there which involves s and p orbitals,
they are
1)sp-hybridisation
2)sp2- hybridization
3)sp3- hybridisation
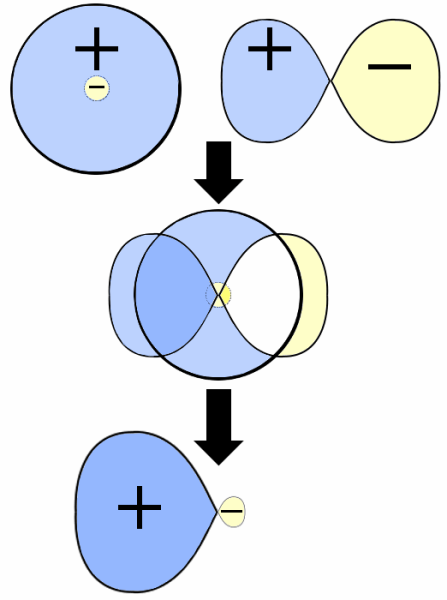
The orbital diagram of hybridisation can be given below


 0
0

