Your Worksheet is Ready
CBSE - Class 11 Chemistry Organic Chemistry – Some Basic Principles and Techniques Worksheet
What is the relationship between the members of following pairs of structures ? Are they structural or geometrical isomers or resonance contributors ?
(a) 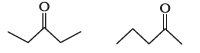
Carbonyl compounds especially ketone undergo reduction to form
Primary alcohol
b.Secondary alcohol
c.Alkane
d.Alkene
Which of the following in eliminations reaction
Chlorination of ethane
b.Hydration of ethene
c.Conversion of but- 1-ene into but-2-ene
d.Dehydration of ethanol
Which of the following carbocation is most stable ?
$(CH_3)_3C. C^+H_2$
b.$(CH_3)_3C^+$
c.$CH_3CH_2C^+H_2$
d.$CH_3C^+H CH_2CH_3$
For the following bond cleavages, use curved-arrows to show the electron flow and classify each as homolysis or heterolysis. Identify reactive intermediate produced as free radical, carbocation and carbanion.
(d) 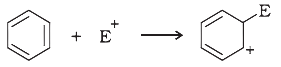
Worksheet Answers