Your Worksheet is Ready
CBSE - Class 12 Chemistry Alcohols Worksheet
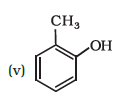
Write IUPAC names of the following compounds:
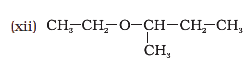
Write IUPAC names of the following compounds:
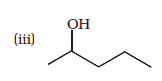
Show how would you synthesise the following alcohols from appropriate alkenes?
Give IUPAC names of the following ethers:
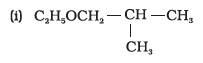
Worksheet Answers