Your Worksheet is Ready
CBSE - Class 12 Chemistry Chemical Kinetics Worksheet
The experimental data for decomposition of \(N_{2}O_{5}\) [\(2N_{2}O_{5} \rightarrow 4NO_{2} + O_{2}\)] in gas phase at 318K are given below:
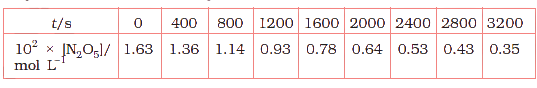
(vi) Calculate the half-life period from \(k\) and compare it with (ii).
In a reaction between \(A\) and \(B\), the initial rate of reaction (\(r_{0}\)) was measured for different initial concentrations of \(A\) and \(B\) as given below:

What is the order of the reaction with respect to \(A\) and \(B\)?
The following results have been obtained during the kinetic studies of the reaction: \(2A + B \rightarrow C + D\)

Determine the rate law and the rate constant for the reaction.
For the decomposition of azoisopropane to hexane and nitrogen at 543 K, the following data are obtained.
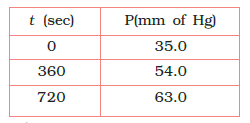
Calculate the rate constant.
The experimental data for decomposition of \(N_{2}O_{5}\) [\(2N_{2}O_{5} \rightarrow 4NO_{2} + O_{2}\)] in gas phase at 318K are given below:
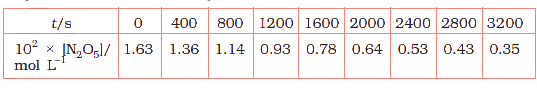
(v) Calculate the rate constant.
The reaction between \(A\) and \(B\) is first order with respect to \(A\) and zero order with respect to \(B\). Fill in the blanks in the following table:

The following data were obtained during the first order thermal decomposition of \(SO_{2}Cl_{2}\) at a constant volume. \(SO_{2}Cl_{2}(g) \rightarrow SO_{2}(g) + Cl_{2}(g)\)
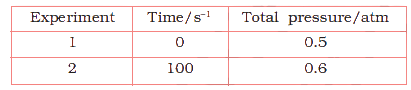
Calculate the rate of the reaction when total pressure is 0.65 atm.
Worksheet Answers