Your Worksheet is Ready
CBSE - Class 9 Science Structure of the Atom Worksheet
Isotopes of an element have
the same physical properties
b.different chemical properties
c.different number of neutrons
d.different atomic numbers.
Composition of the nuclei of two atomic species $X$ and $Y$ are given as under
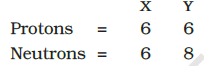
Give the mass numbers of $X$ and $Y$. What is the relation between the two species?
Number of valence electrons in $Cl^-$ ion are:
16
b.8
c.17
d.18
Rutherford’s alpha-particle scattering experiment was responsible for the discovery of
Atomic Nucleus
b.Electron
c.Proton
d.Neutron
Worksheet Answers